Mission
Transforming patients’ lives by revolutionizing healthcare.
Vision
AtomVie is a global leader in the radiopharmaceutical industry dedicated to transforming patients' lives by advancing high quality drugs for the diagnosis and treatment of diseases.
Our Story
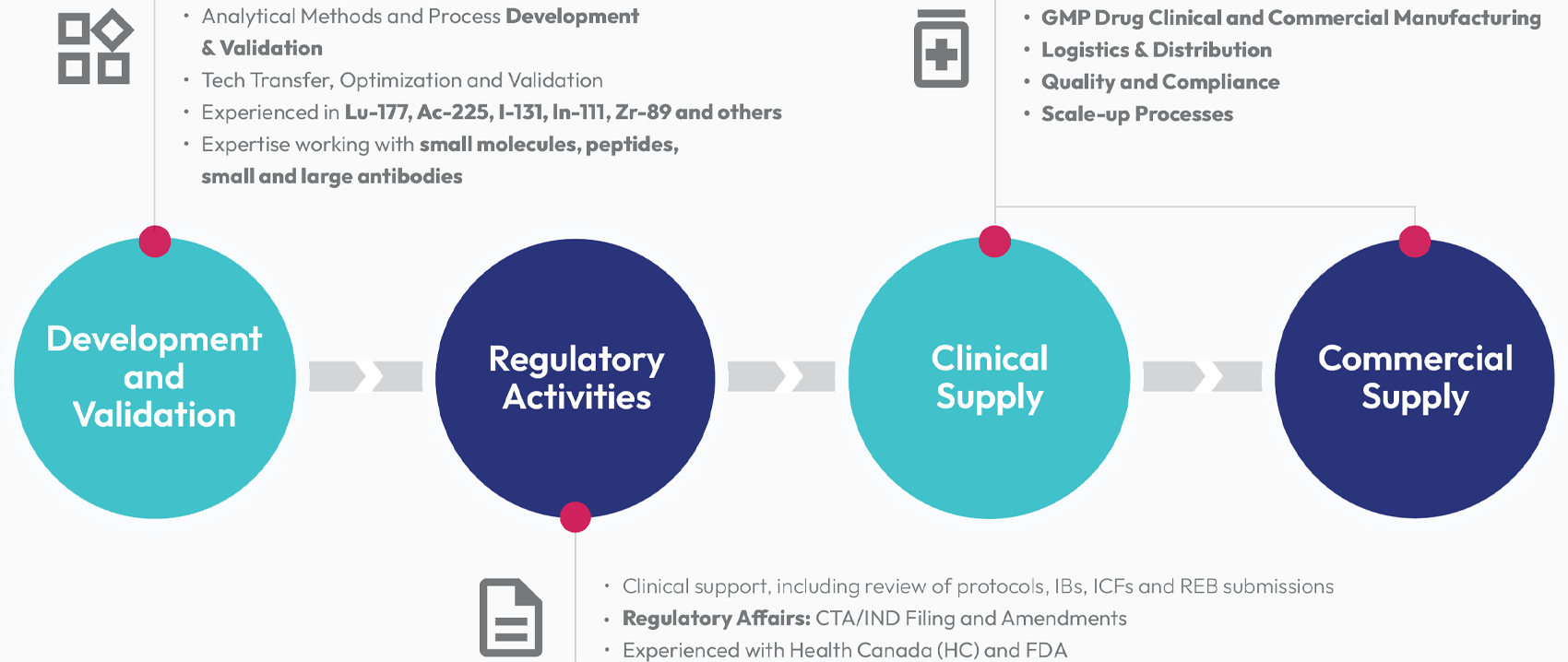
AtomVie Global Radiopharma Inc. (AtomVie) is a global leading Contract Development and Manufacturing Organization (CDMO).
Leveraging over 10 years of expertise in the cGMP manufacturing and global distribution of radiopharmaceuticals by the Centre for Probe Development & Commercialization (CPDC), AtomVie is expanding its manufacturing capacity to offer commercial manufacturing. AtomVie is your CDMO of choice to provide our clients the confidence to successfully execute their clinical development towards commercialization knowing that product supply is reliable, secure and available when and where needed.
AtomVie is currently building and commissioning a new state-of-art purpose built 72,300 sq ft facility, set for operational readiness in H2 2026.
AtomVie offers the full range of scientific, technical, regulatory, quality, logistics and business expertise combined with a specialized infrastructure for the development of your radiotherapeutics from Phase 1 to the commercial marketplace.